Sudden cardiac death is caused by arrhythmias, which is defined as any deviation of the heart beat. Electrical heart disease leading to arrhythmias represents a major public health issue.
The histidine-rich calcium-binding protein (HRC) Ser96Ala variant is known as potential biomarker for sudden cardiac death in patients with a heart muscle disease. HRC is a regulator of sarcoplasmic reticulum (SR) Ca2+ uptake, storage and release. The SR is a membrane bound structure found within muscle cells.
An HRC-Ala96 mouse model using transgenic techniques was developed previously. However, transgenesis has limitations: random positioning, unknown copy number and dependent expression of the inserted DNA. Other issues in the HRC-Ala96 transgenic model arose at the developmental expression of the desired gene dictated by the a-MHC-promoter and the heterologous human gene expression.
To overcome the limitations of HRC overexpression, PolyGene employed a gene targeting knock-in strategy and generated a mouse model carrying an HRC mutation at position 81 (serine 81 to alanine), the homologous region of the human HRC (serine 96 to alanine). This humanized mouse model and subsequent pathophysiological analyses were now published.
(Adapted from Tzimas et al. 2017)
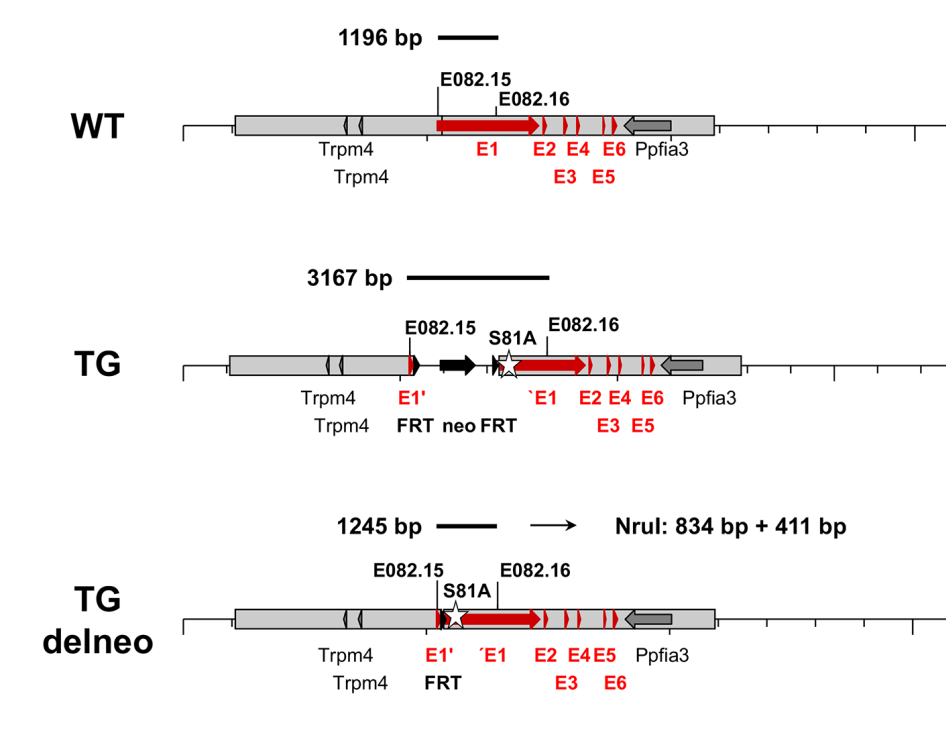
Generation of the HRC-Ser81Ala knock-in mouse model.
Schematic representation of the WT and targeted (TG) HRC-Ser81Ala allele. The targeting vector contains two arms for homologous recombination, separated by a FRT flanked neomycin cassette. The Ser81Ala point mutation is introduced in exon 1 of HRC. Mating of the targeted mice with FLP deleter mice removes the neomycin cassette and results in the generation of targeted and neo-deleted mice.
The generated mutant HRC mouse is physiologically very relevant to the human condition, since the murine endogenous HRC gene remains intact except the specific Ser81Ala mutation. It showed increased mortality in the absence of structural or histological abnormalities, indicating that early death may be arrhythmia-related.
This strongly suggests that indeed Ser96Ala in humans is associated with depressed cardiac function and possible cardiac-related sudden death.
This research was funded by EUTrigTreat, an international consortia founded by the EU, to address both clinical and basic sciences questions about dangerous arrhythmias by translational and interdisciplinary strategies.
>> Online Publication